In a molecular shape, only the atoms are taken into consideration while determining the structure of the molecule.īut in electron geometry, all electron pairs are taken into consideration. 
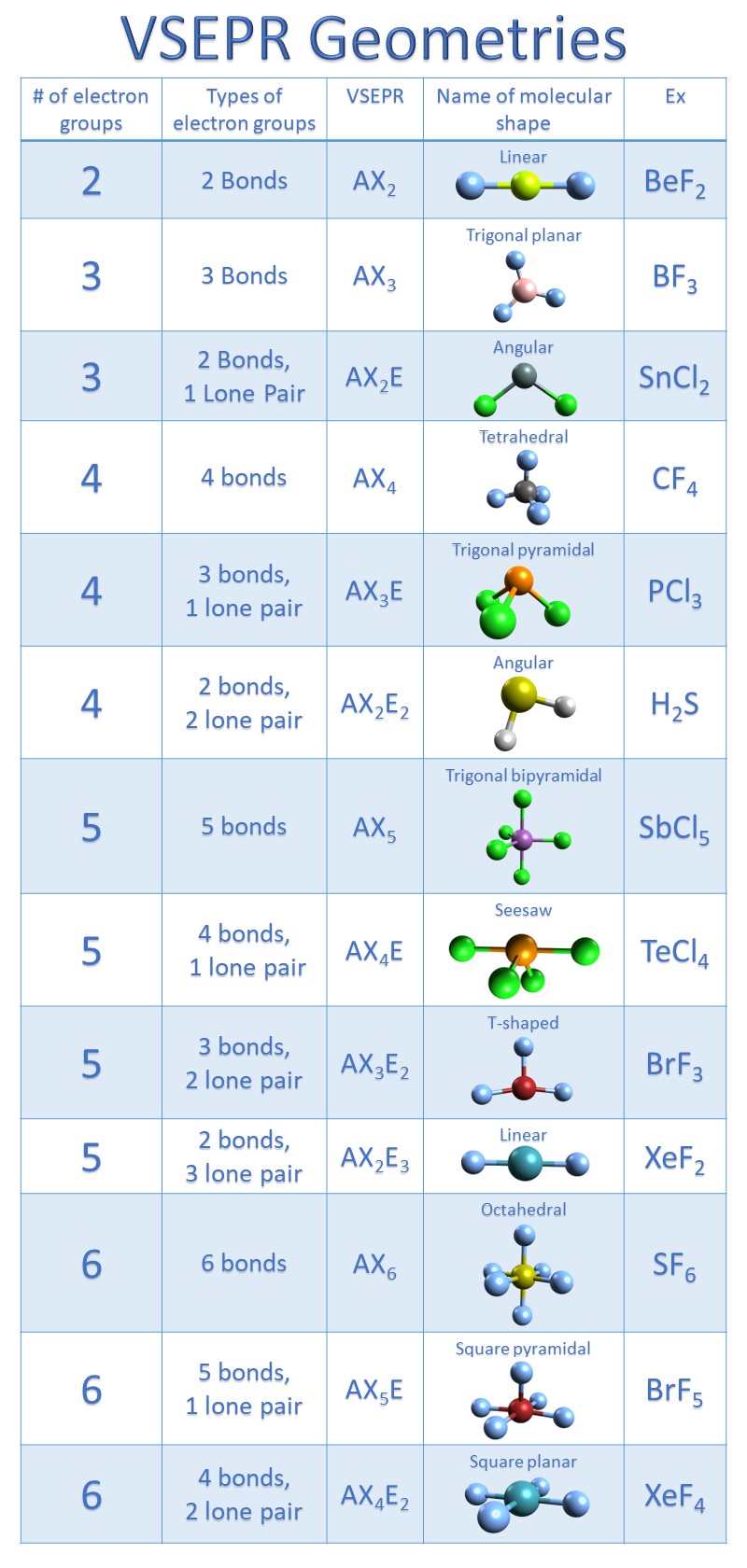
Now there is a clear difference between molecular shape and electronic geometry. The image attached below explains all the above-mentioned concept, So according to the VSEPR chart, H3O+ has trigonal pyramid as its molecular shape and tetrahedral as its electron geometry. (A= central atom, X= bonded atom, E= lone pair on A). The molecular shape of H3O+ is a trigonal pyramid and electronic geometry is tetrahedral.įrom the above chart, we can see that hydronium ion is a AX3E type molecule We can determine the hybridization of a molecule from the VSEPR chart as well, which we will discuss in the next part. Hybridization is generally the mixing of atomic orbitals to form a new hybrid orbital. Thus we can say hybridization of H3O+ is Sp3. In hydronium ion, the central atom is oxygen and it has 6 valence electrons.ģ hydrogen atoms are bonded to oxygen, so the number of the monovalent atoms (M) = 3Īs this is a cationic molecule thus, C = 1 Now let’s find the hybridization of H3O+ using this formula, The hybridization of any molecule can be found using a formula. The hybridization of H3O+ (hydronium ion) is Sp3. 
Chemistry is all about exceptions after all !! Lewis structure rules are almost the same for all molecules, although some exceptions exist here as well. You can calculate the same with the formula given below:. At last, make sure all the atoms are having their lowest possible formal charge.Give multiple bonds if required for fulfilling the octet of the atoms.Keep in mind to start with the electronegative atoms and proceed to the electropositive one. Fillup the octet of the atoms with the remaining electrons.Draw a skeletal structure with single bonds only.Choose a central atom generally the atom with the highest bonding sites.Do take care of +, – signs while calculating. 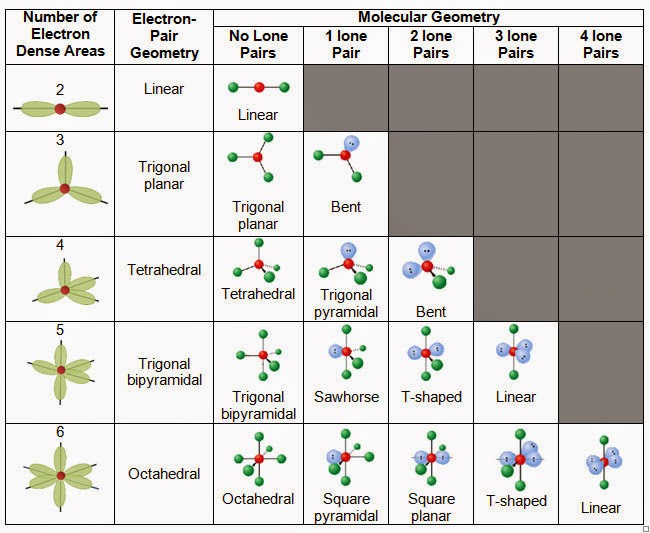
Calculate the total number of valence electrons in the molecule.To simplify the process more for you, I have jotted down the steps below in bullets : There is a common way by which we can draw the lewis structure of any compound. The attached image below shows the lewis structure of hydronium ion Ī lewis structure helps us to find out about the structure of the compound, types, and the number of bonds, physical properties, and how the compound interacts with other compounds.ĭrawing a lewis structure is pretty simple! After that, to complete the lewis structure of H3O+ we have to fill up the octet of atoms with the remaining electrons.Īt the end of drawing the lewis structure of H3O+, we should check that, all the atoms are having their lowest possible formal charge. Next, we need to draw a skeletal structure of H3O+ with single bonds only. In this case, Oxygen is the central atom. Secondly, we need to determine a central atom which is generally the atom with the most available sites for bonding. Thus, the total valence electron is 8 now. + sign indicates losing an electron from the total valence electrons. Now the important point is, not to forget about the + sign. Let’s try to draw the lewis structure of H3O+.įirst of all, we need to calculate the total number of valence electrons present in hydronium ion. So let’s dig deep and know about some interesting information about hydronium ion! We should always try to know the background of any compound before studying any reaction regarding it. Knowing these basics will deepen our knowledge about this ion more. Both organic and inorganic chemistry includes hydronium ion to a large extent.īut before reading the use of this ion in different reactions, we must have knowledge about the basics of this ion, like, lewis structure, geometry, etc. The hydronium ion is used in various reactions and the production of different compounds. As water dissociates into OH- and H3O+ and we can check the pH of the water using a particular process. This ion is used in determining the pH of water. The production of this ion is during the protonation of water The molar mass of the hydronium ion is 19.02 g/mol. Oxonium is a generalized name for all trivalent oxygen cations, so the use of the name hydronium is necessary to identify hydronium ions particularly. If we see the nomenclature of hydronium ion, we get to know that according to the IUPAC nomenclature, hydronium ion can be referred to as oxonium. The cation H3O+ is known as the hydronium ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
